Plastics
Expanded Polyurethane (PUR expanded)
General information
Description
Polyurethanes come in various types and forms. Best known and most produced is PUR foam. This can be hard, soft, and elastic. The latter would be expanded PUR. It exists in an ester and an ether variety. Their degradation differs; esters become tacky and lose their flexibility, ethers will crumble after some time.History
The first polyurethanes were developed in 1937. Around 2010 expanded PUR foam was introduced.Production, Application, Appearance
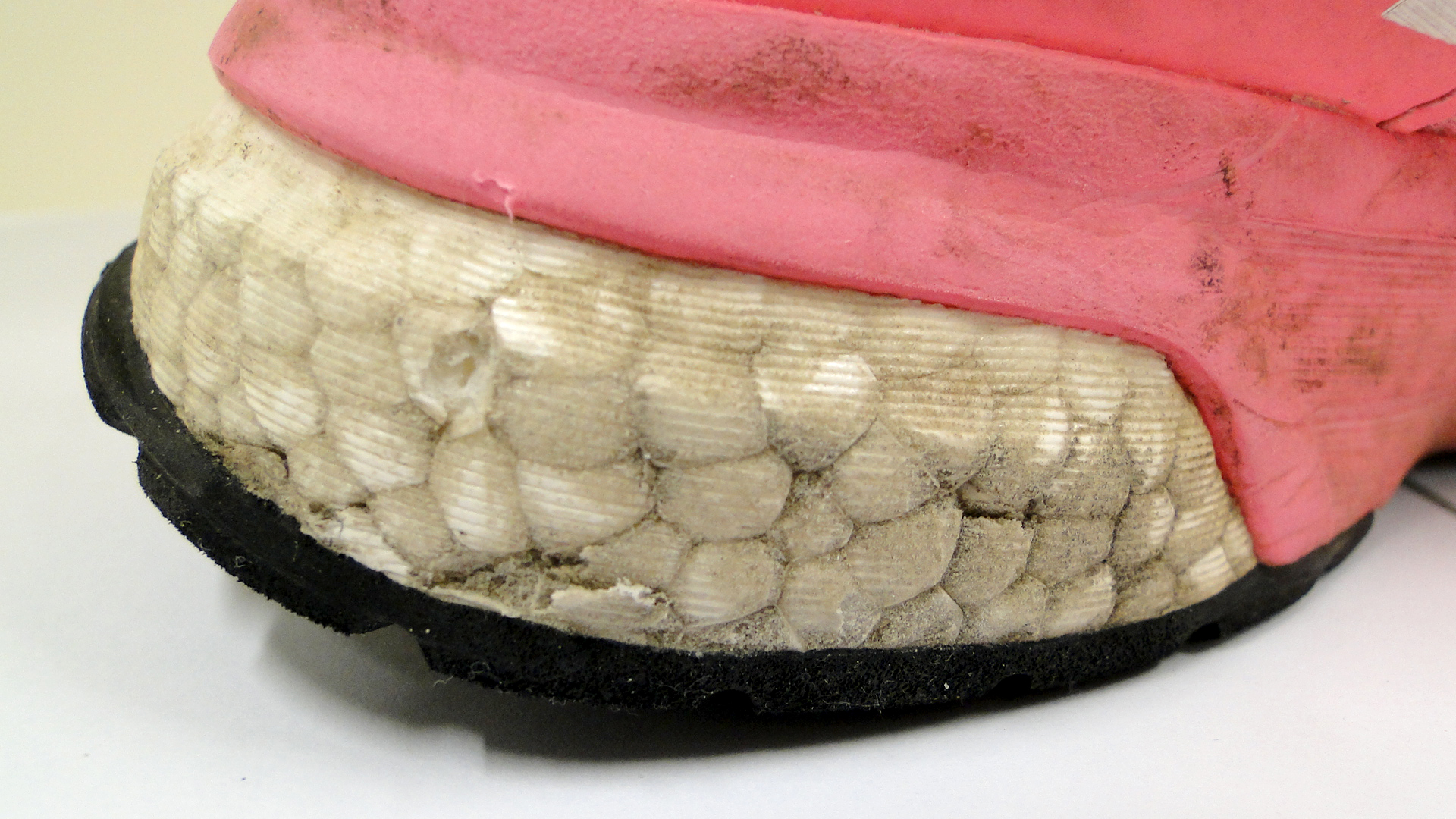
Expanded PUR foam is produced in a similar manner as polystyrene foam: polyurethane granules are expanded with a blowing agent. The two foams therefore have a similar look. The difference between the two is that expanded PUR foam is elastic. The main application is in shoes, the Adidas BOOST shoe being a good example.Properties
Material properties
ThermoplasticDensity: 1.1-1.25 g/cm3; foam: ~0.011 g/cm3
Melting point: 141-157°C
Glass transition temperature: -60 to -19°C
Identification properties
Cell structure (foam): spheresSmell: can have a sweet chemical smell
Touch: no characteristic touch
Sound: no characteristic sound
UV-radiation (when clear): not applicable
Polarizing filters (when clear): not applicable
Degradation
Process
Photo-oxidation, hydrolysis.Details
PUR is considered a problem plastic. Expanded PUR seems to have less problems but still degrades.Symptoms
Discolouration; surface becomes matte; loss of mechanical properties resulting in tears and fractures; surface becomes tacky; efflorescence of white bloom.Susceptibility
Expanded PUR foam is a relatively new product. Little is known about its susceptibility. For the time being the advice is to treat it like other PURs. The susceptibility of flexible polyurethane depends on whether it is a PUR ester or PUR ether. PUR esters are highly susceptible to hydrolysis, at high RH the surface becomes tacky. Ethers have a high susceptibility for photo-oxidation (UV and light) which causes discolouration and crumbling. See appendix for a chemical test to distinguish the two.Ester:
UV-radiation: Medium
Light: Medium
Oxygen/Ozone: Low
Temp: Low
RH: High (setpoint)
Ether:
UV-radiation: High
Light: High
Oxygen/Ozone: Medium
Temp: Low
RH: Medium
Preventive conservation
Recommendations
Ester:UV-RADIATION: keep below 75 µW/lm UV filter for daylight and fluorescent light - reduce intensity
LIGHT: 1 slight change in approx. 30 Mlx.h Moderate light dose - control intensity and exposure time
OXYGEN / OZONE: ambient conditions
TEMP: common indoor conditions 10-30°C; a lower temperature will slow down hydrolysis
RH: 10-30% RH fluctuations: within bandwidth
—
Ether:
UV-RADIATION: keep below 10 µW/lm Exclude UV with filters or no-UV light source
LIGHT: 1 slight change in approx. 1 Mlx.h Limit light dose by reducing intensity and exposure time
OXYGEN / OZONE: lower temperature slows down oxidation
TEMP: common indoor conditions 10-30°C
RH: common museum conditions 40-60% RH fluctuations: setpoint ±10% or ±5% when allowing seasonal fluctations between 35-65%
Other names
- Infinergy
Am I dealing with...
TAGS
- Closed cell structure
- Skin
- Hard
- Ball structure